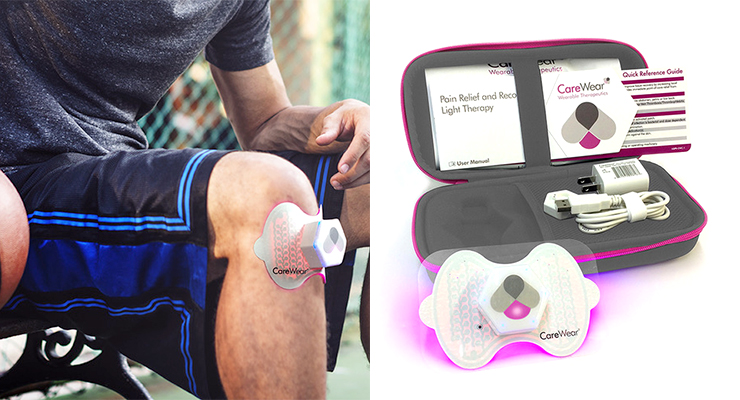
In recent years, medical specialists have realized that the use of light therapy known as PhotoBioModulation can provide specific health benefits. For example, red lights stimulate healing, while blue lights kill bacteria and increase blood flow. The goal is to create systems that can bring all of these benefits to the consumer market, tailoring it to specific needs.
CareWear Corp., Reno, NV, is doing just that, having developed the first wearable, wireless, FDA-registered LED light patches for athletes and consumers, the company notes. Founded in 2017, CareWear is partnering with several experts, including major universities, medical centers and Nth Degree, to create printed electronics systems that enhance health.
With 67 patents and numerous government approvals, CareWear is already successful in a number of markets – CEO Dr. Chris Castel estimates that 50% of sports leagues and branches of the military are using CareWear’s technology to treat soft tissue injuries, and the company sees plenty of new opportunities ahead.
Dr. Castel has been in the medical field for quite some time, having co-founded and built up Accelerated Care Plus to provide services in 4,500 rehabilitation clinics before selling it in 2010, and he sees “wearable therapeutics” as the next great step in treatment, where consumers can reliably use the treatments at home rather than in a clinic.
“We have developed medical device companies, always specializing in physical agent modalities like electric stimulation, ultrasonic therapy and light therapy,” Dr. Castel noted. “These are therapies that are used for pain management and tissue healing commonly performed in physical therapy and rehabilitation clinics.
“We looked at how to take a product that was specifically geared to in-clinic use and take it to the consumer side of the marketplace,” he added. “How do we create a field called wearable therapeutics?”
There are companies that make in-home wearables, but Dr. Castel said the challenge is measuring and delivering the doses accurately. “Lots of people have developed flashlights or masks, which leads to poor compliance and dosage levels that may not produce results. We know what doses work,” he said. “The question is how do we cost it down and keep it effective and easy to use.”
To ensure its ease of use, CareWear developed a philosophy it calls “click, stick and go.”
“By using a thin flexible substrate with a transparent adhesive hydrogel material, our patch optically couples the light to the skin and tissue below it,” Dr. Castel noted. “You stick a patch on your body, attach the controller using its magnetic connector and click to start, then do your own thing without restriction. It turns off automatically and you get the outcome and the dosages you are looking for. The patch is reusable up to 20 times, or as the long as the gel lasts.”
The key differentiator is the delivery system, a highly flexible, conformable substrates that can be manufactured at a low cost.
“Diodes are printed roll to roll with different wavelengths of light,” said Dr. Castel. “We needed flexible substrates for the application of energy into tissue. Initially, we looked at flexible OLED, but it is very expensive to produce, had insufficient optical output and the barrier films were not very good from a shelf life perspective. We began a partnership with Nth Degree; they make the printed light and we handle the medical device side of it.
“We consider that we have developed a platform tech,” he observed. “We have 67 issued patents so far.”
CareWear has earned numerous awards for its technology, including the second place SPIE Startup Challenge Winner in 2018; one of the top 2019 finalists at the 10th IoT/WTInnovation World Cup in 2019; an ISPO Top 5 Startup in sports in 2019; and one of eight companies from CES 2020 chosen by the Consumer Technology Association (CTA) to present at a business pitch contest hosted by AARP.
Having the approval of governments is also strong validation.
“We just recently received Canadian government approval for import into Canada, and are expecting CE marks on our products by June or July,” said Dr. Castel. “They are Class II medical devices in the USA and Class IIA medical devices in Europe. Care Wear’s products are also covered by workman’s compensation in professional sports. We pride ourselves on having taken the hard but correct regulatory route while being backed by published scientific research.”
Key Markets for Wearable Therapeutics
CareWear has some notable successes already and sees numerous potential markets. In particular, CareWear is working with pro sports leagues on the treatment of soft tissue injuries.
“Our product is probably in use by 50% of professional sports teams in the United States,” Dr. Castel reported. “Somebody gets a contusion or sprains an ankle, and blue light stimulates nitric oxide which stimulates oxygen flow, and red light stimulates healing. It’s been a successful market for us.”
CareWear is also looking at a wide range of possibilities.
“We have performed extensive research work at San Diego State University on bacterial kill, and have demonstrated results in clearance of MRSA and acne,” Dr. Castel said. “Our light patches will be used in wound care dressings and for acne treatment, as blue light kills bacteria and red light stimulates healing and reduces pain. We are also developing anti-aging patches for cosmetic use. We are certainly looking at partnership opportunities in the consumer markets. Chronic pain and inflammation such as osteoarthritis is also a key opportunity for our technology. We are actively looking to develop strategic partnership opportunities in select consumer markets.
“The market for wearable therapeutics in PhotoBioModulation is exciting,” Dr. Castel concluded. “Our plan is to bring these products to consumers as well as continuing our development activities for new applications of light therapy.”