David Savastano, Editor10.27.21
Light therapy is a key tool for healthcare. For decades, medical professionals have used natural and LED lights to improve mental and physical health. For example, red lights stimulate healing, while blue lights kill bacteria and increase blood flow. However, as we learn more about the way that light help to relieve pain, new opportunities are emerging.
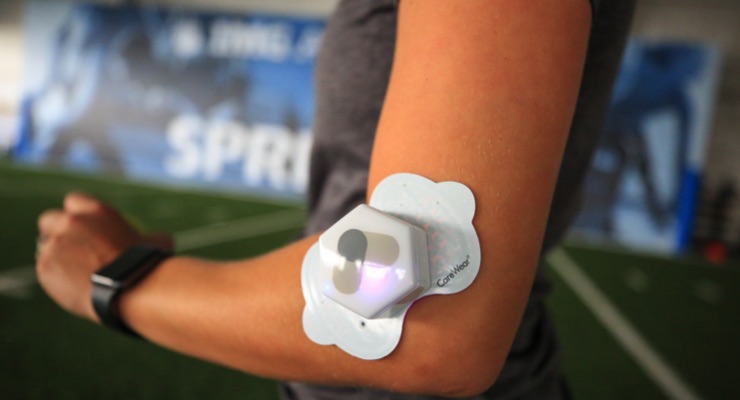
Through its research and development, CareWear Corp., Reno, NV, USA, has become a leader in the field of PhotoBioModulation, or pulsed light technology, to relieve pain. CareWear has been on sale in the US for more than three years and has successfully been adopted by more than 100 professional sporting teams, including teams in the NFL, NBA, MLB and MLS.
While this technology has been used by many sports organizations and the military to treat soft tissue injuries, CareWear Australia sees PhotoBioModulation as a potential aid for arthritis sufferers. Presently, 3.9 million Australians are affected by arthritis, as well as other pain sufferers and athletes.
Dr. Eeshan Kulkarni, CareWear Australia CEO and physicist, said that CareWear’s system uses specific wavelengths of light to trigger chemical responses within the body to relax muscles, stimulate increased nitric oxide production to improve circulation and help relieve pain by accelerating tissue recovery.
“What is really exciting about CareWear is that it is a ‘stick, click and forget’ system that patients can use without assistance, which is really empowering,” Dr. Kulkarni added. “It’s about more than self-management, it’s about giving our users the freedom to take their pain management into their own hands. The CareWear patches are designed to deliver a controlled dose of light therapy and they’re adhesive, so that can used by patients without assistance, which is really empowering for our users.”
CareWear is an ideal example of printed electronics at work. Dr. Kulkarni noted that the CareWear device consists of two parts – the adhesive patch and the wireless controller.
“The patches have adhesive hydrogel on top of printed LEDs, either with quantum dots (the magenta light) or without quantum dots (the blue light),” added Dr. Kulkarni. “Each CareWear patch has 5000+ LED printed lights, developed by NthDegree Technologies.”
Getting the necessary approvals for CareWear’s technology for the treatment of arthritis is the next step. CareWear’s PhotoBioModultation Red Light Therapy is currently being used in trials at the Mater Hospital, and the company reports that early indications show the product has significantly reduced knee pain in trial participants. A larger clinical trial is planned for later this year to confirm the early results.
“As the first wearable, wireless, FDA-registered LED light patches for professionals and consumers, CareWear is an innovative technology, which was challenging and rewarding,” said Dr. Kulkarni. “Our first challenge was developing the printed LED technology and scaling the manufacturing and assembly of the device. Currently, CareWear is in use in trials at the Mater Hospital and we are working to obtain the technology’s CE certification to expand the technology to global markets. When the trials are completed and the CE certificate is obtained, CareWear can then be fully Therapeutic Goods Administration (TGA) approved.”
The launch of CareWear in Australia is a part of CareWear’s global push to bring safe and effective pain relief to all parts of the world.
“At this stage, the CareWear team are investigating expansion opportunities in other markets across Asia and Europe,” added Dr. Kulkarni. “The CareWear Australia team will focus on bringing the technology to patients in Australia and New Zealand.”
Through its research and development, CareWear Corp., Reno, NV, USA, has become a leader in the field of PhotoBioModulation, or pulsed light technology, to relieve pain. CareWear has been on sale in the US for more than three years and has successfully been adopted by more than 100 professional sporting teams, including teams in the NFL, NBA, MLB and MLS.
While this technology has been used by many sports organizations and the military to treat soft tissue injuries, CareWear Australia sees PhotoBioModulation as a potential aid for arthritis sufferers. Presently, 3.9 million Australians are affected by arthritis, as well as other pain sufferers and athletes.
Dr. Eeshan Kulkarni, CareWear Australia CEO and physicist, said that CareWear’s system uses specific wavelengths of light to trigger chemical responses within the body to relax muscles, stimulate increased nitric oxide production to improve circulation and help relieve pain by accelerating tissue recovery.
“What is really exciting about CareWear is that it is a ‘stick, click and forget’ system that patients can use without assistance, which is really empowering,” Dr. Kulkarni added. “It’s about more than self-management, it’s about giving our users the freedom to take their pain management into their own hands. The CareWear patches are designed to deliver a controlled dose of light therapy and they’re adhesive, so that can used by patients without assistance, which is really empowering for our users.”
CareWear is an ideal example of printed electronics at work. Dr. Kulkarni noted that the CareWear device consists of two parts – the adhesive patch and the wireless controller.
“The patches have adhesive hydrogel on top of printed LEDs, either with quantum dots (the magenta light) or without quantum dots (the blue light),” added Dr. Kulkarni. “Each CareWear patch has 5000+ LED printed lights, developed by NthDegree Technologies.”
Getting the necessary approvals for CareWear’s technology for the treatment of arthritis is the next step. CareWear’s PhotoBioModultation Red Light Therapy is currently being used in trials at the Mater Hospital, and the company reports that early indications show the product has significantly reduced knee pain in trial participants. A larger clinical trial is planned for later this year to confirm the early results.
“As the first wearable, wireless, FDA-registered LED light patches for professionals and consumers, CareWear is an innovative technology, which was challenging and rewarding,” said Dr. Kulkarni. “Our first challenge was developing the printed LED technology and scaling the manufacturing and assembly of the device. Currently, CareWear is in use in trials at the Mater Hospital and we are working to obtain the technology’s CE certification to expand the technology to global markets. When the trials are completed and the CE certificate is obtained, CareWear can then be fully Therapeutic Goods Administration (TGA) approved.”
The launch of CareWear in Australia is a part of CareWear’s global push to bring safe and effective pain relief to all parts of the world.
“At this stage, the CareWear team are investigating expansion opportunities in other markets across Asia and Europe,” added Dr. Kulkarni. “The CareWear Australia team will focus on bringing the technology to patients in Australia and New Zealand.”