01.10.20
Schreiner MediPharm will showcase its new Robust RFID-Label, which offers reliable end-to-end functionality from production to final use, at Pharmapack.
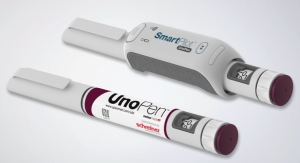
The company also will showcase two products for pharma container integrity: Cap-Lock for sealing prefilled syringes, and Flexi-Cap Prime, which irreversibly indicate any tampering attempt on a vial.
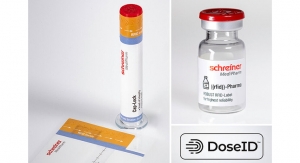
Robust RFID-Label ensures reliable connectivity interaction between users and pharmaceutical manufacturers, as well as communication between products and medical devices, which are important connectivity-centric aspects for pharmaceutical applications. Equipping pharmaceutical products and medical devices with RFID labels results in smart devices that help optimize processes and enhance ease and safety of use. Enhanced robustness and performance of the RFID labels throughout the various processing stages and across the entire supply chain are prerequisites for achieving these objectives.
Schreiner MediPharm’s new Robust RFID-Label consists of a label-integrated RFID tag secured via the label’s special construction. This avoids potential damage to the RFID chip due, for example, to mechanical impact during the manufacturing and handling processes. End-to-end performance of the RFID chip is therefore assured from production to final use – especially on round containers with narrow radii. Schreiner MediPharm customizes the label’s design to suit individual applications and pharmaceutical manufacturing setups.
Cap-Lock and Flexi-Cap Prime have supported the integrity of the supply chain ever since the EU Falsified Medicines Directive came into effect, making tampering with secondary packaging of prescription medicines more difficult.
However, legislation has not yet resolved the issue of ensuring the integrity of primary containers. With Cap-Lock and Flexi-Cap Prime, Schreiner MediPharm is presenting two security concepts that prevent the undetected opening of a primary container, thereby ensuring its integrity. The Cap-Lock cap adapter seals prefilled Luer-lock syringes and indicates if a syringe has been previously opened. While the Flexi-Cap Prime cap label makes any attempt at tampering with a vial irreversibly visible.
The company also will showcase two products for pharma container integrity: Cap-Lock for sealing prefilled syringes, and Flexi-Cap Prime, which irreversibly indicate any tampering attempt on a vial.
Robust RFID-Label ensures reliable connectivity interaction between users and pharmaceutical manufacturers, as well as communication between products and medical devices, which are important connectivity-centric aspects for pharmaceutical applications. Equipping pharmaceutical products and medical devices with RFID labels results in smart devices that help optimize processes and enhance ease and safety of use. Enhanced robustness and performance of the RFID labels throughout the various processing stages and across the entire supply chain are prerequisites for achieving these objectives.
Schreiner MediPharm’s new Robust RFID-Label consists of a label-integrated RFID tag secured via the label’s special construction. This avoids potential damage to the RFID chip due, for example, to mechanical impact during the manufacturing and handling processes. End-to-end performance of the RFID chip is therefore assured from production to final use – especially on round containers with narrow radii. Schreiner MediPharm customizes the label’s design to suit individual applications and pharmaceutical manufacturing setups.
Cap-Lock and Flexi-Cap Prime have supported the integrity of the supply chain ever since the EU Falsified Medicines Directive came into effect, making tampering with secondary packaging of prescription medicines more difficult.
However, legislation has not yet resolved the issue of ensuring the integrity of primary containers. With Cap-Lock and Flexi-Cap Prime, Schreiner MediPharm is presenting two security concepts that prevent the undetected opening of a primary container, thereby ensuring its integrity. The Cap-Lock cap adapter seals prefilled Luer-lock syringes and indicates if a syringe has been previously opened. While the Flexi-Cap Prime cap label makes any attempt at tampering with a vial irreversibly visible.